|
With intermediate formation of hydrogen bonds species. Ammonium chloride (NH 4 Cl) is an ammonium salt which can be used for a variety of applications such as: a catalyst for the one-pot synthesis of 3,4-dihydropyrimidin-2- (1 H )-ones a promoter in the multicomponent synthesis of pyrrolo pyridine-5-one a reagent to improve the hydrolysis rate of magnesium hydride for the. The practitioner's primary taxonomy code is 207Q00000X.Part II: Synthesis and characterization of Cl 2 Dissolve 5.0 g of NO 3 in 50 mL of H 2 O and slowly add concentrated HCl (5-10 mL) until all of the CO 2 gas is evolved. The NPI number of Nh-cl Clinics, Llc is 1487276499 and was assigned on May 2020. Nh-cl Clinics, Llc is a primary care provider established in Kernersville, North Carolina operating as a Family Medicine. This problem has been solved!a (CH 3) 3 N + b HCl = c (CH 3) 3 NH + d Cl 2 Create a System of Equations Create an equation for each element (C, H, N, Cl) where each term represents the number of atoms of the element in each reactant or product.Eight moles of Ammonia and three moles of Dichlorine react to form one mole of Dinitrogen and six moles of Ammonium Chloride Show Chemical Structure Image NH 3 +Cl 2 =N 2 +NH 4 Cl Reaction Type Single Displacement (Substitution) Redox Redox (Oxidation-Reduction) Reaction NH3 + Cl2 = N2 + NH4Cl might be a redox reaction. □ Aprenda Hematologia de forma descomplicada!1. 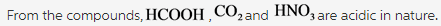
Calculate the following: a) The equilibrium constant at 427☌ c) The standard enthalpy of reaction 4, H- and 459. Padisociation = P xo + PHC) of NH4Cl at 427☌ is 608 kPa but at 459☌ it has risen to 1115 kPa. 
4) Solid ammonium chloride dissociates according to NH Cl (s) →NH, (g)+HCl (g). Best answer Cl - Tetramine Dichoride cobalt (III) chloride ← Prev. answered by Kajal01 (25.8k points) selected by Rajneesh01. 
class-12 Share It On Facebook Twitter Email.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
